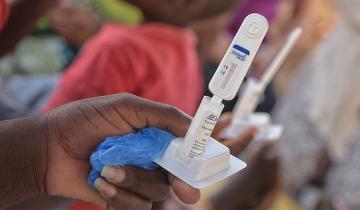
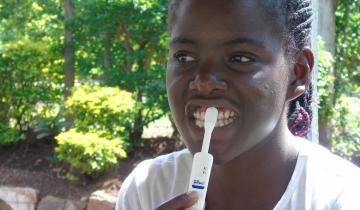
HIV Self-Testing Evidence Program
To gain a better understanding of whether providing HIV self-tests to people would increase HIV testing rates, and who might benefit most from availability of tests, 3ie funded seven pilot interventions and their impact evaluations in Kenya, Uganda and Zambia. The evaluations measure the use of oral HIV self-tests to increase testing rates generally and/or increase the frequency of testing among targeted populations. The results provide robust and actionable evidence on how to safely and effectively promote oral HIV self-testing as an additional testing option.

HIV testing is the first step towards treatment for HIV-positive people, and knowing one’s HIV status is critical for prevention. For these reasons, it is the first target in the UNAIDS 90-90-90 target – that 90 per cent of people living with HIV know their status. However, many countries using current testing options are not on track to meet this target by 2020. HIV self-testing may reduce barriers related to time and travel costs, convenience, confidentiality, health worker interaction and stigma. Implementers and policymakers need more information about how best to make HIV self-tests available to increase testing and knowledge of status, ensure linkage to care, and mitigate the potential for social harms.
Formative research to inform design of pilot interventions
The seven pilot programs covered a range of testing options:
- Offering HIV self-tests to women attending antenatal care to take home and share with their partners in Kenya and Uganda;
- Offering an option of testing methods, including self-tests, to truckers at two roadside wellness clinics in Kenya;
- Alerting truckers and female sex workers, through text messages, of the availability of HIV self-tests at roadside wellness clinics;
- Providing HIV self-tests or coupons to pick up a self-test to female sex workers in Uganda and in Zambia; and
- Adding HIV self-tests as an option in a door-to-door testing intervention to either use in the presence of a health worker, use later, or give to another family member who was not present.
In every study there was an increase in the proportion of people who decided to test.
For more information, please write to info@3ieimpact.org. To receive alerts about call for proposals, please sign up here.
Kenya
3ie collaborated with the Kenyan government and its National AIDS and STI Control Programme. The objective was to generate relevant evidence needed to incorporate HIV self-tests most beneficially into Kenya’s national HIV testing strategy. The ultimate aim is to increase the number of Kenyans who know their HIV status.
We first funded six formative research studies to answer questions about feasibility and acceptability, accuracy, messaging and potential target populations, packaging and labelling, linkage to care, and potential social harms and how to mitigate them. In the second phase, we funded three impact evaluations of pilot programs.
These programs and their evaluation findings have contributed to the government’s efforts to try additional ways of distributing HIV self-tests, including through pharmacies, and by gathering information on willingness to pay and who is willing to pay.
Uganda
3ie funded two impact evaluations in Uganda. The objective was to provide the Ugandan government with the evidence it needed to incorporate HIV self-tests most beneficially into Uganda’s national HIV testing strategy. The ultimate aim is to increase the number of Ugandans who know their HIV status. The evaluation findings have contributed to the government’s current assessment of ways to roll out HIV self-testing.
Zambia
3ie supported one formative research study and two impact evaluations. The goal was to provide policy-relevant evidence that the ministries of health and community development and social services can use to determine whether and how to implement and scale up the availability of HIV self-tests and related services, such as access to counselling and linkage to care. Zambia is hosting another large HIV self-testing initiative (HIV Self-Testing Africa, or STAR) and an HIV combination prevention study (HPTN 071).